Table of Contents
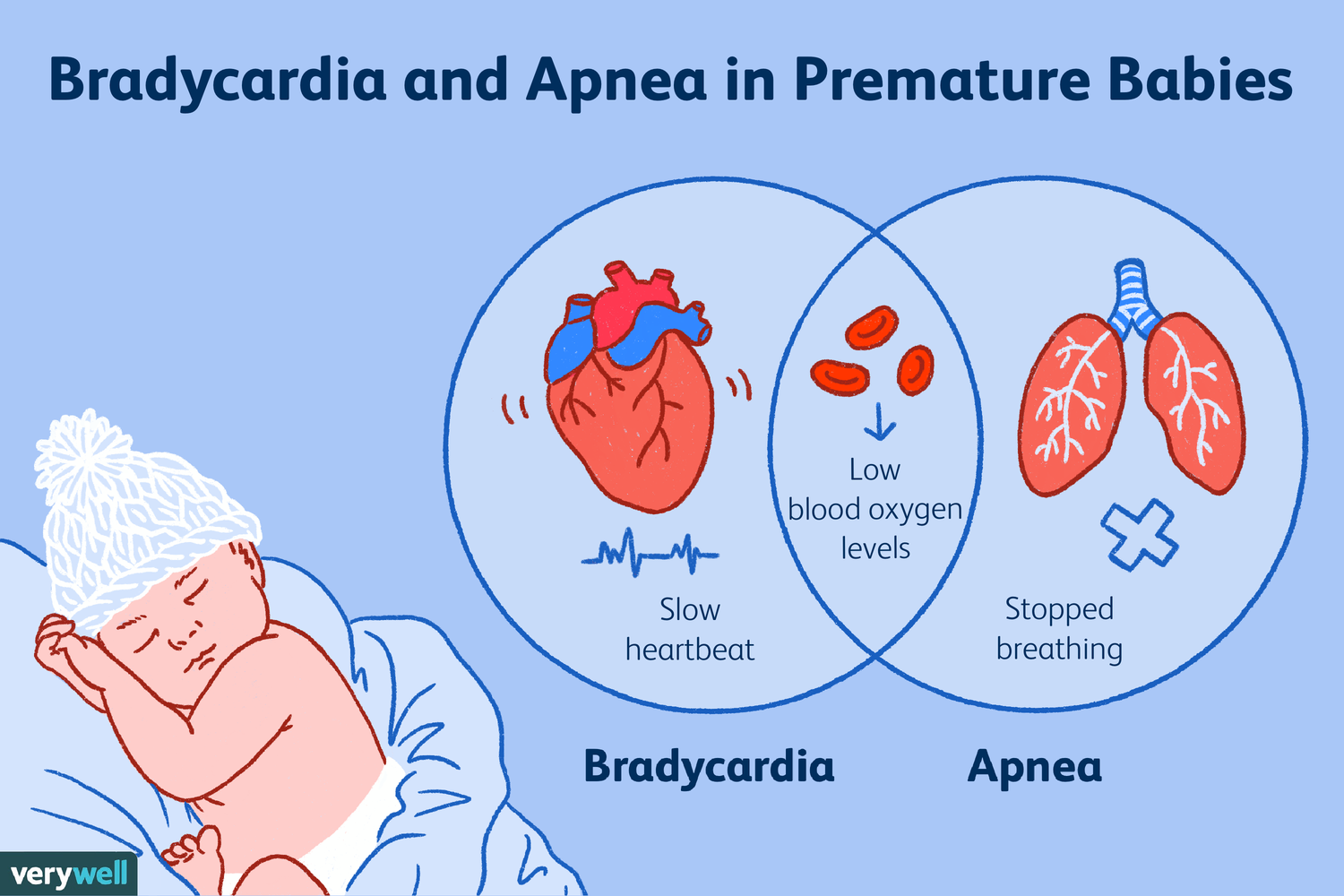
Apnea of prematurity (AOP) is a common condition where infants born before 37 weeks gestation experience breathing pauses lasting 15-20 seconds or more, often accompanied by slow heart rates (bradycardia) or low oxygen levels. It stems from an immature central nervous system and usually resolves on its own by 37–40 weeks postmenstrual age. Based on Cloherty and Stark’s Manual of Neonatal Care (9th Edition) and relevant pediatric algorithms and Nelson’s, here is a comprehensive summary of Apnea of Prematurity (AOP).
1. Definition and Classification
- Definition: Apnea is the cessation of airflow. It is considered pathologic (an apneic spell) when:
- Absence of airflow lasts 20 seconds or longer.
- It is shorter than 20 seconds but accompanied by bradycardia (heart rate < 100 bpm) or hypoxemia (cyanosis or $SpO_2$ < 85–80%).
- Classification:
- Central: Total absence of inspiratory efforts (no diaphragmatic activity).
- Obstructive: Inspiratory efforts persist, but airflow is blocked, usually at the pharyngeal level.
- Mixed: A combination where airway obstruction precedes or follows central apnea. Most spells in preterm infants are mixed.
2. Incidence, Onset, and Duration
- Incidence: Inversely related to gestational age (GA). It occurs in essentially all infants < 28 weeks’ GA and about 25% of those < 34 weeks’ GA.
- Onset: Typically begins 1 to 2 days after birth. If spells do not occur within the first 7 days, AOP is unlikely to develop later unless triggered by other factors.
- Duration: Usually ceases by 36 to 37 weeks’ postmenstrual age (PMA) in infants born at $\ge 28$ weeks, but frequently persists beyond term for those born more prematurely.
3. Pathogenesis (Underlying Mechanisms)
- Developmental Immaturity: The primary cause is an immature central respiratory drive in the brainstem.
- Sleep State: Spells are more frequent during active (REM) sleep, which is the predominant sleep state in preterms and is characterized by irregular breathing.
- Chemoreceptor Response: Preterm infants have a decreased ventilatory response to increased $CO_2$ and may respond to hypoxia with hypoventilation rather than sustained hyperventilation.
- Reflexes: Apnea can be triggered by stimulation of the posterior pharynx (e.g., vigorous suctioning), lung inflation, or fluid in the larynx.
- Airway Mechanics: Poor muscle tone can lead to airway obstruction, especially during neck flexion or if there is nasal obstruction.
- Note on GER: While gastroesophageal reflux is common in preterms, studies have not demonstrated an association between GER and AOP frequency.
4. Differential Diagnosis (Evaluation)
Apnea in a term infant or a “sick” preterm infant is always abnormal and requires looking for secondary causes:
- Infection: Sepsis, meningitis, or necrotizing enterocolitis.
- Metabolic Disorders: Hypoglycemia, hypocalcemia, or electrolyte imbalances (hyponatremia).
- Neurologic: Intracranial hemorrhage (IVH), seizures, or birth asphyxia.
- Impaired Oxygenation: PDA (Patent Ductus Arteriosus), anemia, or pneumonia.
- Drugs: Maternal medications (magnesium, narcotics) or drug toxicity (e.g., phenobarbitone).
5. Management and Treatment
- Monitoring: All infants < 35 weeks’ GA should be monitored for at least the first week. Monitor heart rate and $SpO_2$ in addition to respiration.
- Immediate Action: Respond to the infant, not the monitor alarm. Most spells respond to tactile stimulation. If the infant is unresponsive, use bag-and-mask ventilation.
- Positioning: Avoid extreme neck flexion or extension. Prone positioning may reduce apnea by stabilizing the chest wall.
- Pharmacotherapy (Methylxanthines):
- Caffeine Citrate: The drug of choice due to its long half-life (once-daily dosing), high therapeutic index, and lack of need for routine level monitoring.
- Dosing: Loading dose of 20 mg/kg (10 mg/kg caffeine base), followed by a maintenance dose of 5–10 mg/kg daily.
- Benefits: Reduces spells, the need for mechanical ventilation, and the risk of Bronchopulmonary Dysplasia (BPD).
- Respiratory Support:
- nCPAP (4–6 $cm H_2O$): Reduces mixed and obstructive spells by maintaining end-expiratory lung volume.
- NIPPV: May be attempted if CPAP fails.
- Other Considerations:
- Blood Transfusion: May be considered if the hematocrit is < 25–30% and spells are frequent despite caffeine.
- GER Treatment: Pharmacologic treatment of reflux (e.g., H2 blockers) is not recommended to treat AOP and may be harmful.
6. Discharge and Follow-up
- Discharge Criteria: Infants should be free of significant apnea for 5 to 7 days after stopping caffeine.
- Caffeine Offset: Because caffeine effects remain for up to a week, the “countdown” to discharge typically starts several days after the last dose.
- Recurrence: Recurrent apnea can be triggered by viral illness, anesthesia, eye examinations, or immunizations. These infants should be monitored closely until at least 44 weeks’ PMA.
- SIDS: A history of AOP does not increase the risk of Sudden Infant Death Syndrome (SIDS).