Table of Contents
Diamond-Blackfan anemia (DBA) is a rare congenital blood disorder characterized by the failure of the bone marrow to produce red blood cells. It usually presents in infancy and is classified as a congenital pure red cell aplasia. DBA is notable for its genetic basis, variable physical malformations, and lifelong management challenges.
Key facts
- Onset: Typically within the first year of life
- Genetic cause: Mutations in ribosomal protein genes
- Inheritance pattern: Autosomal dominant (most cases de novo)
- Prevalence: About 5–7 per million live births
- Treatment options: Corticosteroids, chronic transfusions, or stem cell transplantation
Pathophysiology
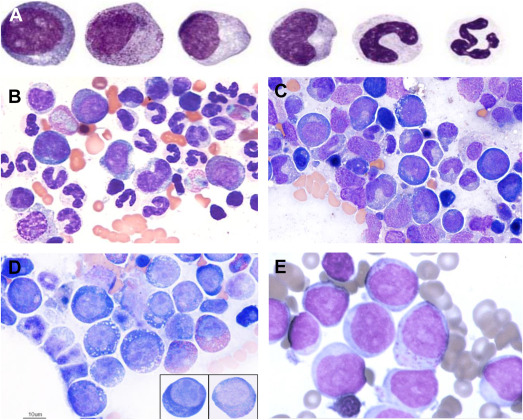
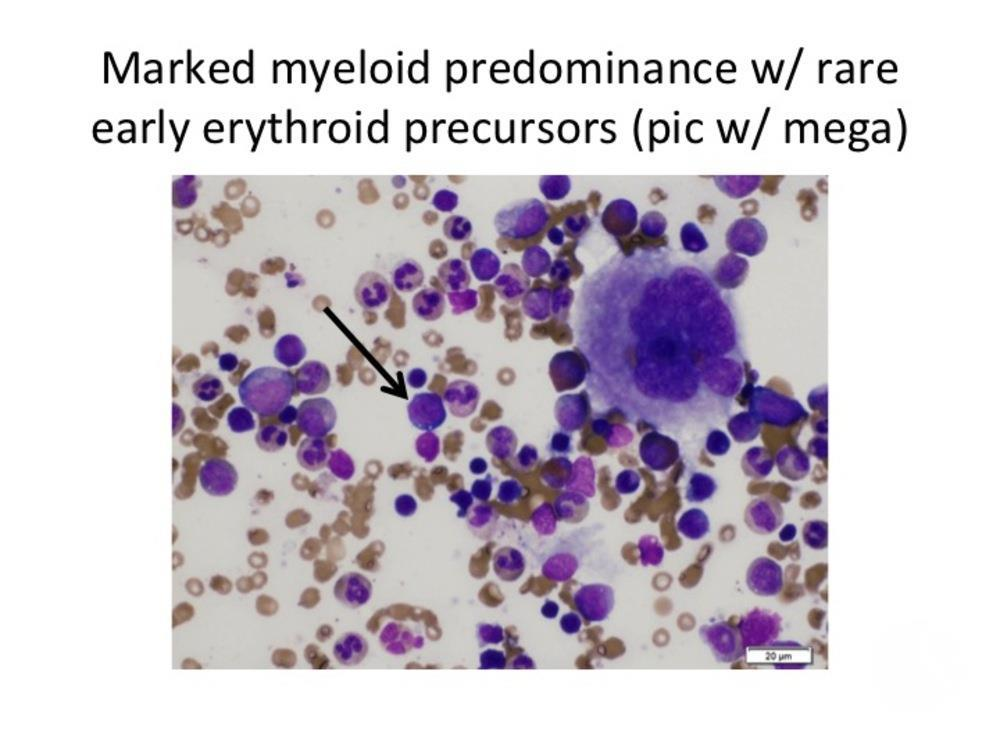
Diamond-Blackfan anemia arises from mutations that impair ribosome biogenesis, leading to defective erythroid progenitor development. The bone marrow becomes selectively deficient in red cell precursors, while white cells and platelets remain normal. Most cases involve mutations in genes encoding ribosomal proteins such as RPS19, RPL5, or RPL11, disrupting protein synthesis and cellular growth.
Clinical features
Infants with DBA commonly present with pallor and anemia. Physical anomalies are present in about half of cases, including craniofacial abnormalities, thumb or limb malformations, and heart or kidney defects. Growth retardation and an increased lifetime risk of malignancies such as leukemia and osteogenic sarcoma are recognized complications.
Diagnosis
Diagnosis combines hematologic findings—macrocytic anemia, reticulocytopenia, and normal marrow cellularity except for absent red cell precursors—with genetic testing for ribosomal protein gene mutations. Elevated erythrocyte adenosine deaminase (eADA) activity is a common biomarker.

Management and prognosis
Initial treatment often involves corticosteroids to stimulate red cell production. Patients unresponsive to steroids may require regular transfusions with iron chelation therapy to prevent overload, or hematopoietic stem cell transplantation as a potential cure. Advances in genetic understanding have improved prognosis, but lifelong monitoring remains essential due to treatment complications and cancer risk.